 This method is widely used when the compound of interest is a liquid. Into another in order to form a solid compound whose melting point serves as a second data A derivative is the conversion of one functional group on your molecule Making a Derivative: OximeĪn historical method to identifying a product was by making a derivative of your compound and While your reaction mixture warms to room temperature,Īnswer the following questions with your partner. While your reaction mixture cools, answer the following Given your answer to P2, propose the rest of the mechanism by which benzaldehyde Once all of the acid mixture has been added, remove the 250 mL flask from the iceīath and allow it to sit for 30 minutes warming to room temperature. Slowly add the mixture of nitric acid/sulfuric acid to the 250 mL flask using a glassĥ. Add benzaldehyde to the sulfuric acid-only 250 mL flask and stir vigorously withĤ. Propose the mechanism for what’s happening in step 2 above:ģ. Leave both Erlenmeyer flasks in the ice bath until both are iceĬold. Add 4 mL of sulfuric acid and the nitric acid to a 150 mL Erlenmeyer flask and place Add 12 mL of sulfuric acid to a 250 mL Erlenmeyer flask and place the flask in an iceĢ. 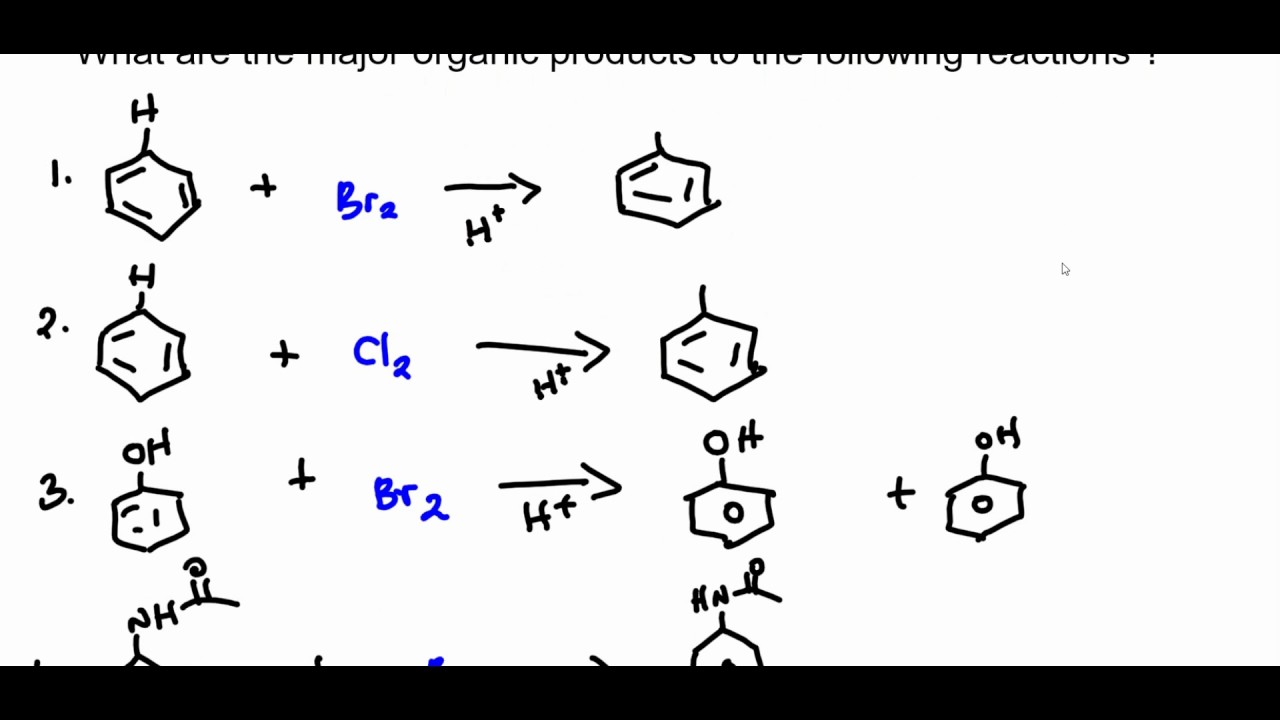
In your lab notebook, prepare an entry for running a reaction using the reaction schemeĬompound Amount Mol Equiv. Obtain the following items in no particular order: Purification will be the main focus next week. Isolation andĬharacterization of the crude products will be required today while Time is available during the reaction to stop and think more criticallyĪbout what’s happening at the molecular level. Substitution will be run over a 30 minute reaction time. Will you get whatįormation of nitrobenzaldehyde via electrophilic aromatic In this lab, you will be tasked withĭeriving a hypothesis for what the major product will be, running the experiment, collecting yourĭata, and then deciphering what the data supports relative to your hypothesis. Position of the nitration is not indicated in the scheme. The following reaction is an electrophilic aromatic substitution of benzaldehyde. Organic REActivity Electrophilic Aromatic Substitution The Reaction Scheme: Nitration of benzaldehyde Educational Research: Competencies for Analysis and Applications (Gay L.Interpersonal Communication (Kory Floyd).Bursten Catherine Murphy Patrick Woodward) Chemistry: The Central Science (Theodore E.Forecasting, Time Series, and Regression (Richard T.Brunner and Suddarth's Textbook of Medical-Surgical Nursing (Janice L.Business Law: Text and Cases (Kenneth W.Give Me Liberty!: an American History (Eric Foner).Principles of Environmental Science (William P.Biological Science (Freeman Scott Quillin Kim Allison Lizabeth).The Methodology of the Social Sciences (Max Weber).Civilization and its Discontents (Sigmund Freud).Techniques DE Separation ET Analyse EN Biochimi 1.SEC-502-RS-Dispositions Self-Assessment Survey T3 (1).School-Plan - School Plan of San Juan Integrated School.I am doing my essay on the Ted Talk titaled How One Photo Captured a Humanitie Crisis https.Leadership class, week 3 executive summary.Module One Short Answer - Information Literacy.Essentials of Psychiatric Mental Health Nursing 8e Morgan, Townsend.NHA CCMA Practice Test Questions and Answers. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
